Investigational New Drugs: FDA Has Taken Steps to Improve the Expanded Access Program but Should Further Clarify How Adverse Events Data Are Used | U.S. GAO
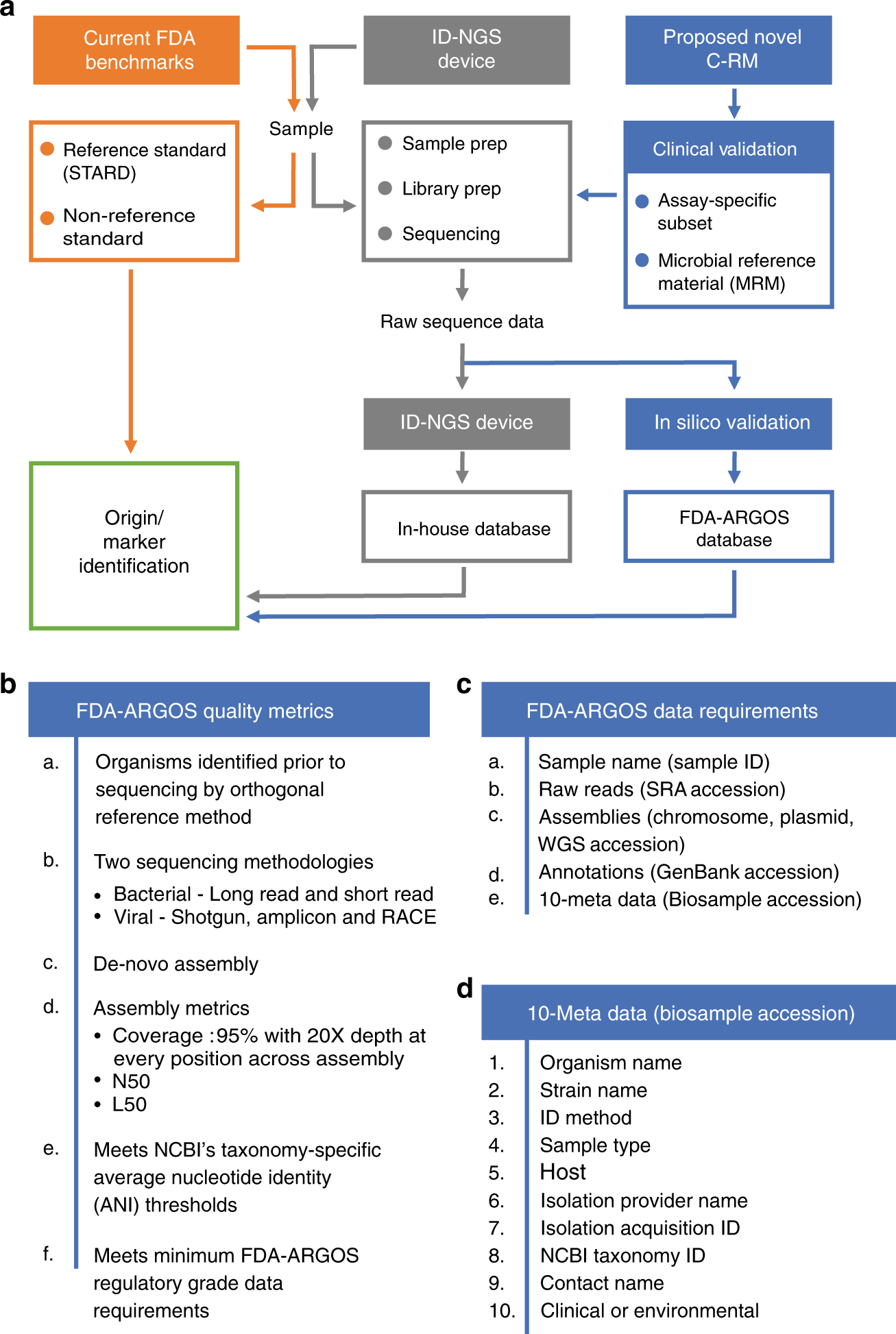
FDA-ARGOS is a database with public quality-controlled reference genomes for diagnostic use and regulatory science | Nature Communications

Transparency advocates win victory for public access to clinical trial data | Center for Science in the Public Interest

How Often Are Drugs Made Available Under the Food and Drug Administration's Expanded Access Process Approved? - McKee - 2017 - The Journal of Clinical Pharmacology - Wiley Online Library

Vague FDA policies on adverse event data are keeping patients from accessing investigational drugs | Fierce Healthcare

19 Printable certificate of analysis fda Forms and Templates - Fillable Samples in PDF, Word to Download | PDFfiller
![PDF] IDAAPM: Integrated database of ADMET and adverse effects of predictive modeling based on FDA approved drug data PDF] IDAAPM: Integrated database of ADMET and adverse effects of predictive modeling based on FDA approved drug data](https://i1.rgstatic.net/publication/303977127_IDAAPM_Integrated_database_of_ADMET_and_adverse_effects_of_predictive_modeling_based_on_FDA_approved_drug_data/links/5900b6774585156502a00f65/largepreview.png)
PDF] IDAAPM: Integrated database of ADMET and adverse effects of predictive modeling based on FDA approved drug data

U.S. FDA on Twitter: "FDA launches interactive database with crucial information about life-saving, HIV drugs available for purchase under the PEPFAR program as part of ongoing mission to empower the public through